Copper Dime: Debunking the Myths
What do you know about a copper dime? Possibly, the coin should look brown; the surface shows no silver tone.
In reality, no regular U.S. dime was ever issued fully in copper for circulation. If a dime appears copper, there is a reason — and it is usually wear, damage, or a mint error. Here is how to tell the difference.
Why the Myth Persists
The idea of a copper dime spreads fast. A brown surface looks unusual. That alone convinces many people.
Online listings make the confusion worse. Normal wear is described as rare. Headlines use dramatic language, and claims repeat without verification.
Older silver dimes add another layer of misunderstanding. Before 1965, dimes were 90% silver. After 1965, the composition changed. Some collectors mix these eras when talking about metal content.
Now let’s address the most common claims directly:
“Any brown dime is rare.” → False
“No mint mark means copper.” → False
“Magnet test alone proves authenticity.” → False
“Found in change means valuable.” → Rare
But remember: color is not evidence. Absence of a mint mark is not proof. One simple test does not confirm an error.
In numismatics, rarity must be verified. Weight, diameter, edge structure, and professional authentication matter. What else should we know about the dime?
What Were Dimes Made Of?
Understanding composition removes most confusion.
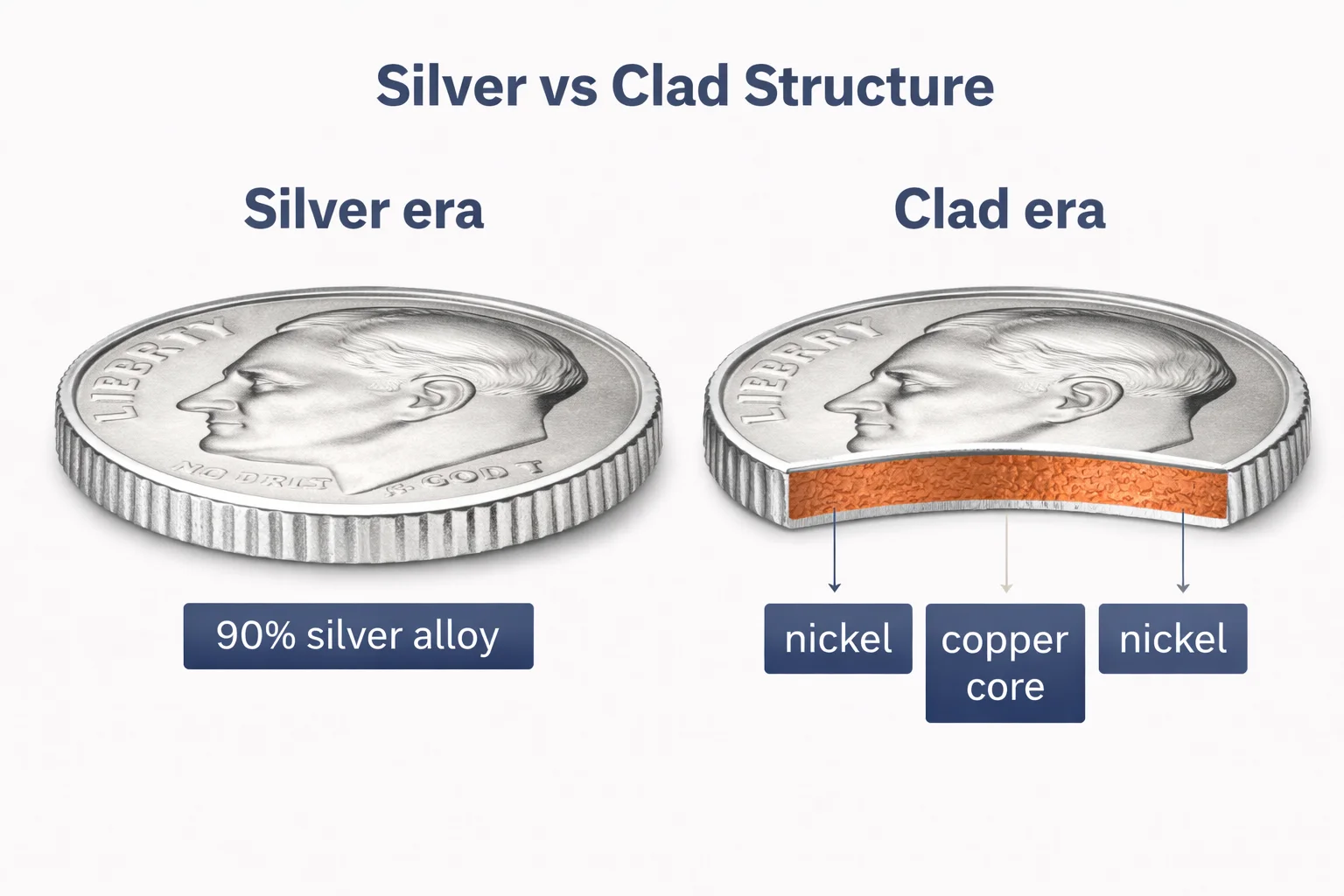
1796–1964: Silver Era
For more than a century, U.S. dimes were made of 90% silver and 10% copper. They were not pure silver. They were not pure copper.
These coins contain copper as part of the alloy, but they are silver coins.
1965–Present: Clad Era
Starting in 1965, the composition changed. Dimes became copper-nickel clad coins.
They consist of:
Copper core
Nickel outer layers
This design creates the familiar silver color. The edge reveals a thin copper stripe. But no circulating dime was issued as a fully copper coin.
Why Some Dimes Look Copper
A dime can appear brown for several reasons.
Heavy wear removes the nickel surface. The copper core becomes visible.
Environmental damage can darken metal. Soil exposure, moisture, or chemicals change surface tone.
Heat can alter color. Fire damage sometimes turns coins dark or reddish.
In some cases, the outer clad layer separates. When one side loses the nickel coating, the coin looks copper on that side.
Appearance does not equal composition.
How Real Copper Dimes Appear
But what about real copper dimes? There are legitimate scenarios where a dime may truly be copper in composition. These are mint errors, all of them are extremely rare, and most of them are certified.
Struck on the Wrong Planchet
This is the most realistic explanation.
A dime design can be struck on a cent blank. That blank is copper alloy.
When this happens, the coin shows:
Smaller diameter
Weight around 3.11 grams
Copper color throughout
Incomplete or weak design
These coins are off-metal errors. They occur when a blank intended for another denomination enters the striking press.
They are collectible and must be authenticated.
Off-Metal Strike
This term describes a coin struck on a planchet made for another denomination or metal type.
Signs include:
Unusual weight
Incorrect diameter
Different edge structure
Surface color is inconsistent with standard issues
Authentication is essential. Without certification, claims remain speculation.
Experimental or Pattern Pieces
During transition periods, the Mint sometimes tested metal compositions. These patterns rarely entered circulation.
They are documented. They appear at major auctions. They are not common pocket finds.
Most people will never encounter one in daily change.
How to Check a If You Found a Possible Copper Dime
If you believe you found a copper dime, just check it according to the steps below.
Step 1: Weigh the Coin
A standard clad dime weighs about 2.27 grams. A cent planchet weighs about 3.11 grams. If the weight matches a normal dime, it is not struck on a cent blank.
Step 2: Measure the Diameter
A standard dime has a 17.91 mm diameter. A cent planchet is larger. And size differences are often visible without tools.
Step 3: Inspect the Edge
A clad dime shows a layered structure. A true off-metal piece struck on a cent blank will not show that layered look. It will appear solid copper.
Step 4: Check if the Hue Is not a Damage
Remember that dimes have a copper core, and most scratches, corrosion, or plating can create a copper-like appearance. And these are not mint errors or valuable pieces.
Step 5: Consider Professional Certification
Only grading companies such as PCGS or NGC can confirm off-metal status.
Before submitting, collectors sometimes use Coin ID Scanner to confirm year, denomination, and basic specifications. The app helps organize data and compare standard technical parameters. It supports identification but does not replace laboratory authentication.
Verification is the key difference between rarity and misunderstanding.
Market Value: When Is a Copper Dime Valuable?
A genuine off-metal dime can attract strong collector demand.
Value depends on:
Confirmed error type
Year of issue
Condition
Eye appeal
Certification
Minor surface issues bring little premium.
Documented wrong-planchet strikes can range from several hundred dollars to several thousand. Dramatic examples in high grades bring the strongest prices. The market rewards rarity that can be proven.

Final Verdict
How often do real off-metal dimes appear? Very rarely.
The United States Mint maintains strict quality control. Planchet errors occur, but they represent a tiny fraction of production.
Most alleged copper dimes are simply worn coins. Collectors who specialize in mint errors examine thousands of coins to find one authentic off-metal strike.
If you suspect an unusual coin:
Do not clean it.
Do not alter it.
Record weight and diameter.
Compare with verified examples.
Seek professional opinion.
Cleaning or polishing destroys evidence. It removes diagnostic surfaces. It reduces collector interest. Original condition preserves credibility.
Prices depend on rarity and documentation. Dramatic mistakes attract stronger bidding. Common damaged coins do not qualify. The difference between a damaged clad dime and a true copper off-metal piece is measurable.
Taking all the above, remember the rule: understanding metal composition prevents false expectations. The hobby rewards knowledge, not assumptions.

